Pfizerleaks: Concerning patterns in the fertility and developmental study RN9391R58?
Many observations such as swelling, malformations, abortions, stillborns, total litter loss, and weak pups occurred predominantly or exclusively in vaccinated rats
The November 1, 2022, release of Pfizer trial documents by Public Health and Medical Professionals for Transparency includes the final report for study no. RN9391R58, “A Combined Fertility and Developmental Study (Including Teratogenicity and Postnatal Investigations) of BNT162b1, BNT162b2 and BNT162b3 by Intramuscular Administration in the Wistar Rat” (pdf file). Arguably this study is (was) relevant towards assessing the safety of Pfizer’s mRNA vaccine in pregnant women and newborn babies. On pages 8 and 13, we can see that the study was planned by June 2020, completed primarily through July-September 2020, and results were reviewed by the study director at Charles River Laboratories in France on December 10, 2020, almost two years ago.
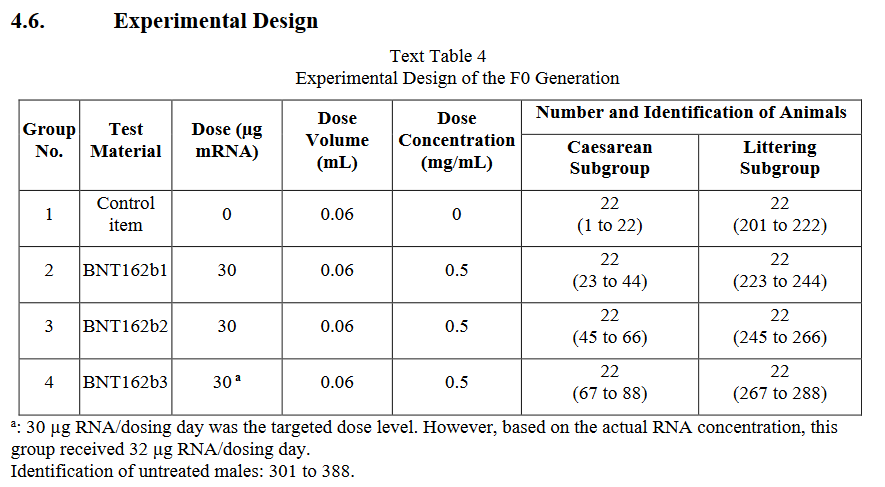
Now, I will say that I find animal testing unethical and am hesitant to write about it. But I do find human testing even more unethical, in particular if it is conducted at a global scale. Thus, let’s have a critical look at this Pfizer/BioNTech-funded study. They used Wistar lab rats divided into four groups, a control group and three vaccine groups to test three BioNTech vaccine candidates, BNT162b1, BNT162b2, and BNT162b3. Each group was further divided into a caesarean subgroup to study embryo development and a littering subgroup to study live pup development, with 22 female animals in each subgroup for a total of 178 female rats. The animals in the vaccine groups received four mRNA injections, two prior to mating (with untreated males) and two during gestation, while the controls received a saline solution.
I will spare you (and myself) the other details on the test setup and procedures, and jump right into the results from Section 8 (page 30 onwards) of the final report. A first set of results concerns the first generation of rats (mothers). Section 8.2 Mortality indicated that three animals died prematurely, all in the vaccine groups. The researchers consider these unscheduled death incidental with reference to historical control data. Two of the three deaths were in animals removed from the study after total litter loss. To put these into context, the report refers to cases of total litter loss in a mere two out of 18 other studies conducted between 2015 and 2019. That does not look very convincing to me.
Additionally, they refer to the zero deaths in the BNT162b2 vaccine group to dispute a relation to any of the candidate products, but fail to acknowledge the zero deaths in the control group or the impact of low probabilities in conjunction with the small number of test subjects. In other words, if we were to find a 66% chance (two out of three vaccine groups) that 1 in every 44 human mothers could lose their babies, we would hopefully conduct a few more safety tests on the drug they received, no?
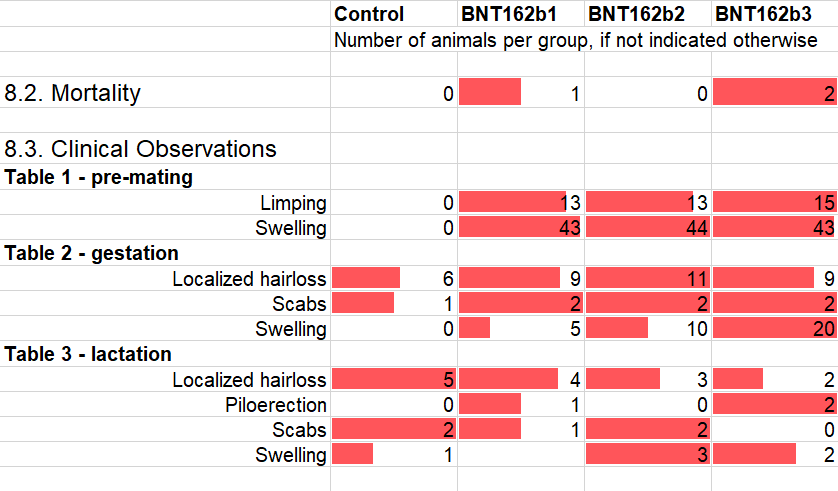
Under Section 8.3 Clinical Observations and the associated Tables 1-3, I selected only a few types of observations that had multiple occurrences and/or showed a marked pattern. I included several observations with occurrences in the control group but there are quite a few that are primarily or exclusively befalling the vaccinated animals.
In the following Section 8.9 Fetal Examinations, literally ALL malformations and other anomalies and “variations” affected the fetuses of vaccinated mothers. The numbers are small to be sure, but the pattern is striking nonetheless.
The last section to be discussed here is Section 8.10 Delivery and Litter Data. The vaccinated mothers in the b1 and b3 vaccine groups experienced in the order of TWICE as much pre-birth loss as the unvaccinated (12.2% to 13.8% compared to 6.8%). Here is the researchers’ interpretation of this observation: “…but the differences were not biologically meaningful and the value remained consistent with the historical control data range (from 5.1% to 13.6%) for pivotal studies, so the difference was considered to be incidental.” However, the control group is near the low end of the historical range, while two vaccine groups are at the high end, with one of them actually exceeding the historical maximum. Nothing to see here?
A similar picture emerges around pup viability. Live-births from vaccinated mothers are 3.3% to 4.8% lower than from unvaccinated mothers, which the researchers dismiss as “marginally lower”. To put this into context, 95% live birth compared to 98% live birth in 1,000 pregnancies means 30 dead babies. For reference, in 2021, Canada had around 99% live birth among a total of 370,858 births according to Statistics Canada’s table 13-10-0428-01. A drop by 3% would mean 11,126 additional stillborn babies per year.
The report also blames the poor performance of the vaccine groups on the two instances of total litter loss. The researchers are clearly biased in viewing these instances as incidental rather than drawing any conclusions from the fact that they did occur in vaccinated groups and not in the unvaccinated animals.
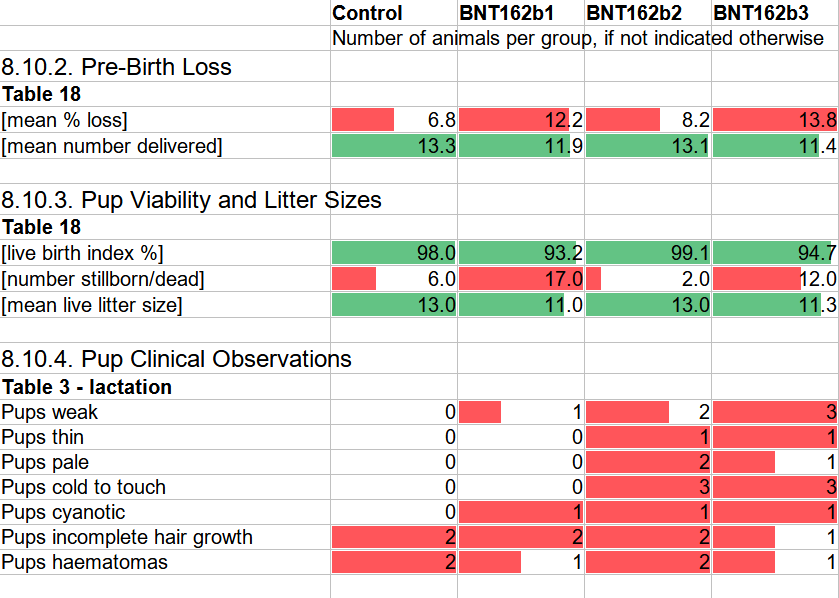
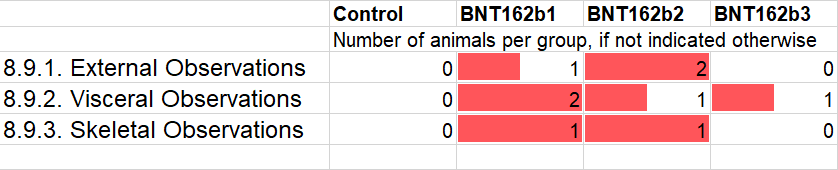
Interestingly, the b2 vaccine group is not mentioned in the text of Sections 8.10.2 and 8.10.3, and I almost missed its metrics as they are only included in the associated Table 18. The values are in fact comparable to the control group, and in terms of live birth index (99.1%) and number of stillborns (2) outperform the controls. It should be noted that BNT162b2 is the candidate product that was selected and approved, e.g. through Health Canada’s interim order on December 9, 2020. I wonder whether these pregnancy outcome data were known to Pfizer/BioNTech when they selected b2 over b1 or b3.
The health of the Wistar rat pups again gives reason for concern, as five out of seven indicators shown above are shifted towards issues in all vaccine groups. Again, I have selected indicators where I subjectively noticed a pattern based on multiple observations across multiple groups, whether there was a clear distinction between the control and the treatment arms or not. Specifically, these remaining observations are consistent between the vaccine candidate groups. If anything, the b1 group looks better than b2 and b3. This element does not give me much confidence in the selected b2 vaccine candidate, in particular since I am assuming that most of the ingredients of these concoctions are identical. In fact, the drug regulators should have known before approval how similar or different the BNT162b2 cocktail is to the two other, more deadly b1 and b3 recipes.
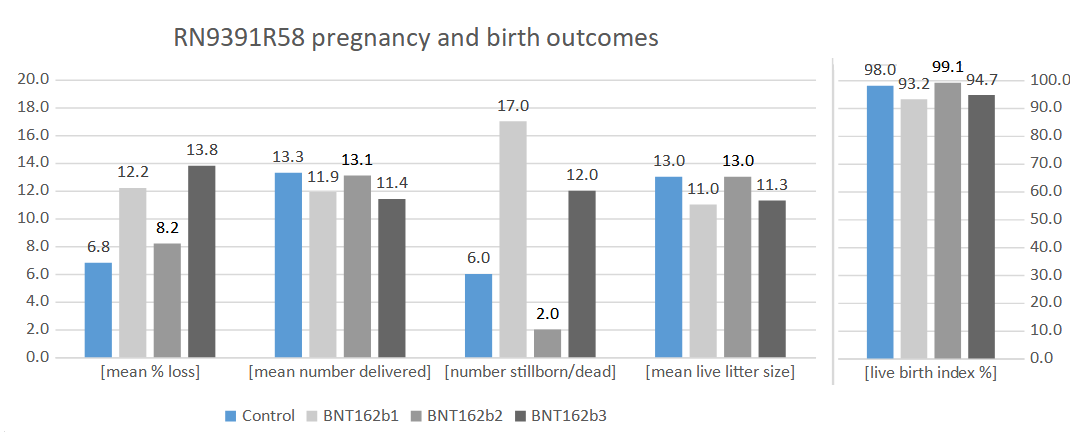
The summary statistics from Sections 8.10.2 and 8.10.3 are a bit harder to read than the case counts in the other observations. Here is another way to look at these data with the blue bars representing the unvaccinated control group and the three shades of grey being the b1, b2, and b3 vaccine groups. The mean% loss of pups and the number of stillborn pups show the shockingly higher incidence in the b1 and b3 groups as compared to the control group. Even the 21% (8.2% compared to 6.8%) higher mean loss of fetuses in the b2 group should be viewed as a safety signal. The mean number of pups delivered, mean live litter size, and live birth% show the concerningly reduced performance of the same two out of three vaccinate groups.
I can believe that the differences between observations were not statistically significant, yet the researchers seem to intentionally minimize or ignore the directional trend of these differences between the unvaccinated and vaccinated groups in many categories of observations. The results were obtained in December 2020 around the time that health authorities around the globe were approving the mRNA products. A copy of the report, albeit with a smaller total page number, is posted on the Australian Government’s Therapeutic Goods Administration web site with a file name suggesting a freedom-of-information request. That document is heavily redacted for references to the b1 and b3 vaccine candidates, and even the number of products tested was removed in this earlier release.

One would hope that the Australian Government along with Health Canada, the US Food and Drug Administration, and other regulators examined this study before they approved the injections for pregnant women. However, to this day, His Majesty’s Government of the United Kingdom states in the decision summary of the December 2020 Pfizer/BioNTech approval (page updated November 9, 2022), that “A combined fertility and developmental study (including teratogenicity and postnatal investigations) in rats is ongoing.” This is the exact title of the study discussed here, so are we to believe that the UK approved the shots and pushed them onto pregnant women without looking at these data?
Since we are approaching the two-year mark of study completion, we might also ask how the global 5-billion human experiment of 2021-2022 worked out overall? There are worrisome reports about declining birth rates in a number of countries. Until I get a chance to dive into this issue, I refer to a post by Dr. Joshua Guetzkow from July 2022, “Springtime for Stillbirths in Germany”, which also includes references to other countries’ recent vital statistics and their critical interpretation.